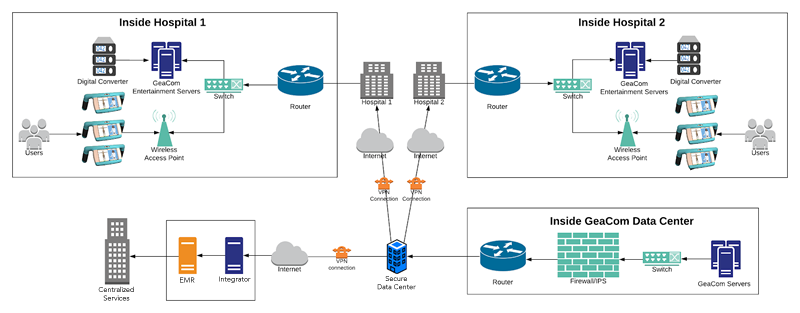
The fastest, most affordable equity innovation is GeaCom’s promise (and literally the only proven equity solution). The unmatched, blazing innovation pace is only realized due to purpose built, medical grade CITE tools that include hardware, user interfaces, operating system, specialized server stacks and direct content development. A full feature rich foundation for rapid market driven innovation is proving unparalleled. Without further preamble, here are the 2019 breakthrough advancements:
CITE Engagements
GeaCom’s greatest value is the proven capability to more effectively engage anyone despite language, literacy, gender, culture, age and more. During 2019 the uncontested world leading engagements expanded by more than 4 fold.
Virtual Health
A new feature for rapidly transitioning from CITE engagements to on-system (live video) physician engagements and back, seamlessly was incorporated. Patients and staff now engage and advance care more effectively, even in the ‘waiting room’. This feature is also used for care provided out in the community where mental health assessments or general engagements identify populations with greater need and bring the care team directly to them (in their homes, at work or in the community), at the moment of need.
Fast Track
In concert with seamless live engagements, GeaCom and its medical advisors added identifiers to determine which patients could be “fast tracked” to care and discharge without even being roomed. This feature enables physicians, in facility or out, to see patients who can be quickly engaged, assisted and discharged without further impact on resources. Estimates from medical research indicate that as many as 40% of ER and Urgent Care visits can be fast tracked, saving time, money and resources while improving patient experience.
Mesh Network Functionality
Integrating new, highly responsive technology in a burdened infrastructure can be expensive, daunting and additive but with the new Mesh functionality the entire integration is run on a self generated network. No wiring, no additional network hooks, no routers and load balancing hardware; just place Phrazer/Kitsune and it handles the rest.
MRM Enhancements
Multi-path Realtime Messaging is at the core of CITE functionality. Previous iterations focused on one-to-one benefits (one patient, one staff listener) but now the expanded MRM adds custom messages to individual staff members. This feature directs staff to most efficient performance, process adherence and constant patient awareness that is highly specific to the individual staff member. Additional MRM use of EMR, alert boards and the caregiver Kitsune bring vast possibilities to this breakthrough feature. The most stunning advancement of the new MRM is that it can engage staff offsite as well for realtime, live updates enabling fast track and virtual engagements.
PEPS
The new Patient Entertainment Portal and Services feature adds a whole new interface set that patients LOVE. In the patient’s own language and appropriate to their age and culture, this interface provides self guided health engagements and avast array of entertainment.
Tethering and Network Sharing
Often medical systems have poor network or patients can’t access their cell networks and now in these cases, Phrazer/Kitsune can create custom private networks for patient’s friends and family to access and use for both PEPS and Internet services.
Screen Casting
This new feature provides the ability to ‘cast’ what is on a Phrazer/Kitsune screen to one or multiple other screens. You can cast PEPS to a large screen or host group video conferences from Phrazer/Kitsune. The casting also makes it possible to send live updates to all patients or a sub-group by casting a master screen. This feature also opens possibilities for controlling content to waiting room TV screens and hosting group trainings.
OHA Expansions
The Open Handset Alliance feature enables a secure, isolated access for incorporating Android or iOS applications, natively, onto Phrazer/Kitsune. GeaCom has added live translation partners ‘Apps’ such as LanguageLine, CyraCom and Google translate solutions. Also added are games, podcasts, books and specialty staff reference tools. The OHA means you can run your favorite Apps on the most secure, fastest and only qualified platform without any additional coding or costs.
PPV and Revenue (sponsorships and advertisements)
GeaCom certified as a JP Morgan Chase payment gateway which enables Pay-Per-View and other commerce features on Phrazer/Kitsune. This feature enables patients to access more premium services. Also incorporated are sponsorship and advertising interfaces so users may choose free services while the managing system experiences increased revenue.
Faster EMR connections
Our partners and GeaCom have worked diligently to ensure faster, lower cost and more expensive EMR connectivity. During 2019 the cost has dropped by more than 50% while the time required to integrate decreased by up to 80%.
OpenGL Engagements
After years of development GeaCom is ready to unveil the new high speed, high response 3D interfaces. Fully interactive and dynamically adaptive human models, matching the patient, are now available for countless CITE engagements to further enhance the look, feel and value of the experience.
Process Improvement and Adherence
With the new Mesh Network, casting, new caregiver Kitsune and enhanced MRM, vast horizons in process improvement, staff adherence and realtime patient/staff/system coordination have opened. Realtime touch to all assets can now coordinate with AI to create best performance facilities.
Staff Competency Development
Phrazer/Kitsune staff training and competency engagements are now available for everything from cultural competency to policy and procedures and CMEs to CDC notifications. For new on-boarding staff to reinforcement for established staff; staff competency on the CITE platform educates, confirms and assures everyone is on the ‘same page’.
K-12 and Community Engagements
After 3 years of in market research, performance and breakthrough results, the new CITE Althing was released with rich K-12 and community interfaces and features. Bridging best of class hardware, CITE engagements and education with community services is proving to be the most powerful community health and services technology ever conceived. Seamless transitions from pediatric to community to shared decision making interfaces; all community focused and owned is a new breakthrough of CITE.
OTA Updating
Secure “Over The Air updating” is fully enhanced for realtime evolution of Phrazer/Kitsune engagements. No IT overhead, no delays and even use of the new mesh function enables realtime updates, fast, lightweight and reliably. With innovation as a cornerstone OTA brings high speed innovation to reality.
What Really Matters
Never loosing focus on equity in engagements, the Civil Rights benefits, innovation and being the best of class platform remains GeaCom’s core philosophy and during 2019 it was well maintained and enhanced.



 Wait times are significantly longer
Wait times are significantly longer